Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2025-10-03
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2025-10-21
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2025-12-04
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2026-01-26
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2025-08-20
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2026-01-14
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2025-11-30
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2026-02-06
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2026-02-06
Test MethodHPLC + Mass
Spectrometry
LaboratoryBioAnalytical Labs, Inc.
Test Date2026-02-06

SageBioLabs sells research-grade peptides exclusively for laboratory and scientific research purposes. These compounds are not intended for human consumption.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | Tirzepatide |
| Lot Number | LOT-2026-001-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.5% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 4813.5 g/mol | 4813.30 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |

\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | Retatrutide |
| Lot Number | LOT-2026-002-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.9% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 6758.22 g/mol | 6758.02 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |

\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
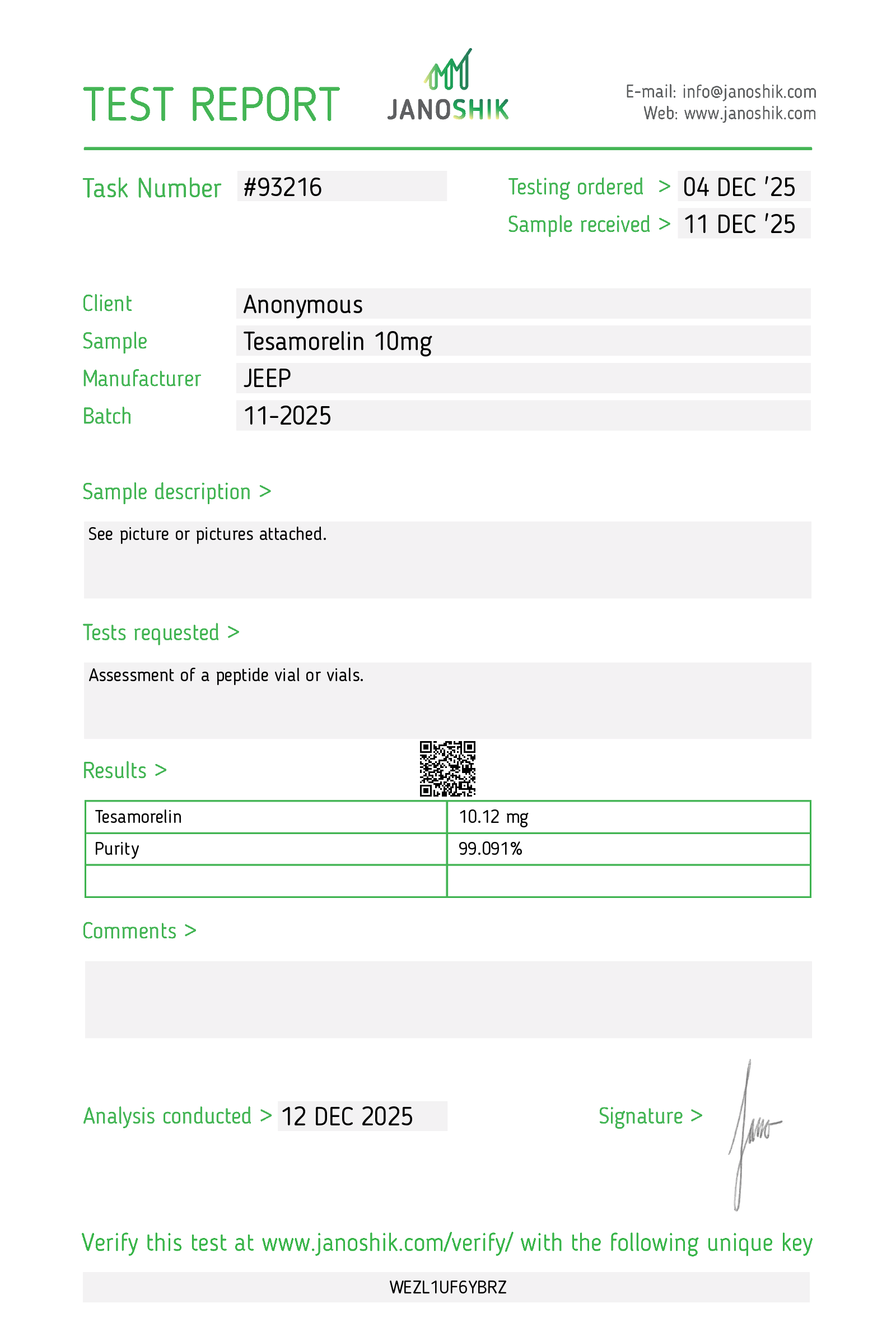
| Product Name | Tesamorelin |
| Lot Number | LOT-2026-006-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.2% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 5135.8 g/mol | 5135.60 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |
\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | BPC-157 |
| Lot Number | LOT-2026-003-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.1% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 1419.56 g/mol | 1419.36 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |

\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | BPC-157 |
| Lot Number | LOT-2026-003-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.1% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 1419.56 g/mol | 1419.36 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |

\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | GHK-Cu |
| Lot Number | LOT-2026-003-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.1% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 1419.56 g/mol | 1419.36 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |

\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | GLOW 50 |
| Lot Number | LOT-2026-003-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.1% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 1419.56 g/mol | 1419.36 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |

\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
Certificate of Analysis · Independent Third-Party Testing
Testing conducted independently. Results verified by dual analyst review. Certificate ID: CVP-2026-002
| Product Name | Melanotan |
| Lot Number | LOT-2026-003-A |
| Manufacturing Date | January 10, 2026 |
| Test Date | January 15, 2026 |
| Expiry Date | January 2028 |
| Form | Lyophilized Powder |
| Storage | −20°C, protected from light & moisture |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Purity (HPLC) | ≥ 98.0% | 99.1% | ✓ PASS |
| Single Max Impurity | ≤ 1.0% | 0.4% | ✓ PASS |
| Total Impurities | ≤ 2.0% | 0.9% | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Molecular Identity (MS) | Confirmed match | Confirmed | ✓ PASS |
| Molecular Weight | 1419.56 g/mol | 1419.36 g/mol | ✓ PASS |
| Test Parameter | Specification | Result | Status |
|---|---|---|---|
| Bacterial Endotoxin (LAL) | < 2 EU/mg | < 1 EU/mg | ✓ PASS |
| Sterility (Visual) | No visible contamination | Compliant | ✓ PASS |
| Moisture Content (KF) | ≤ 6.0% | 3.2% | ✓ PASS |
\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.

Certificate of Analysis · Independent Third-Party Testing
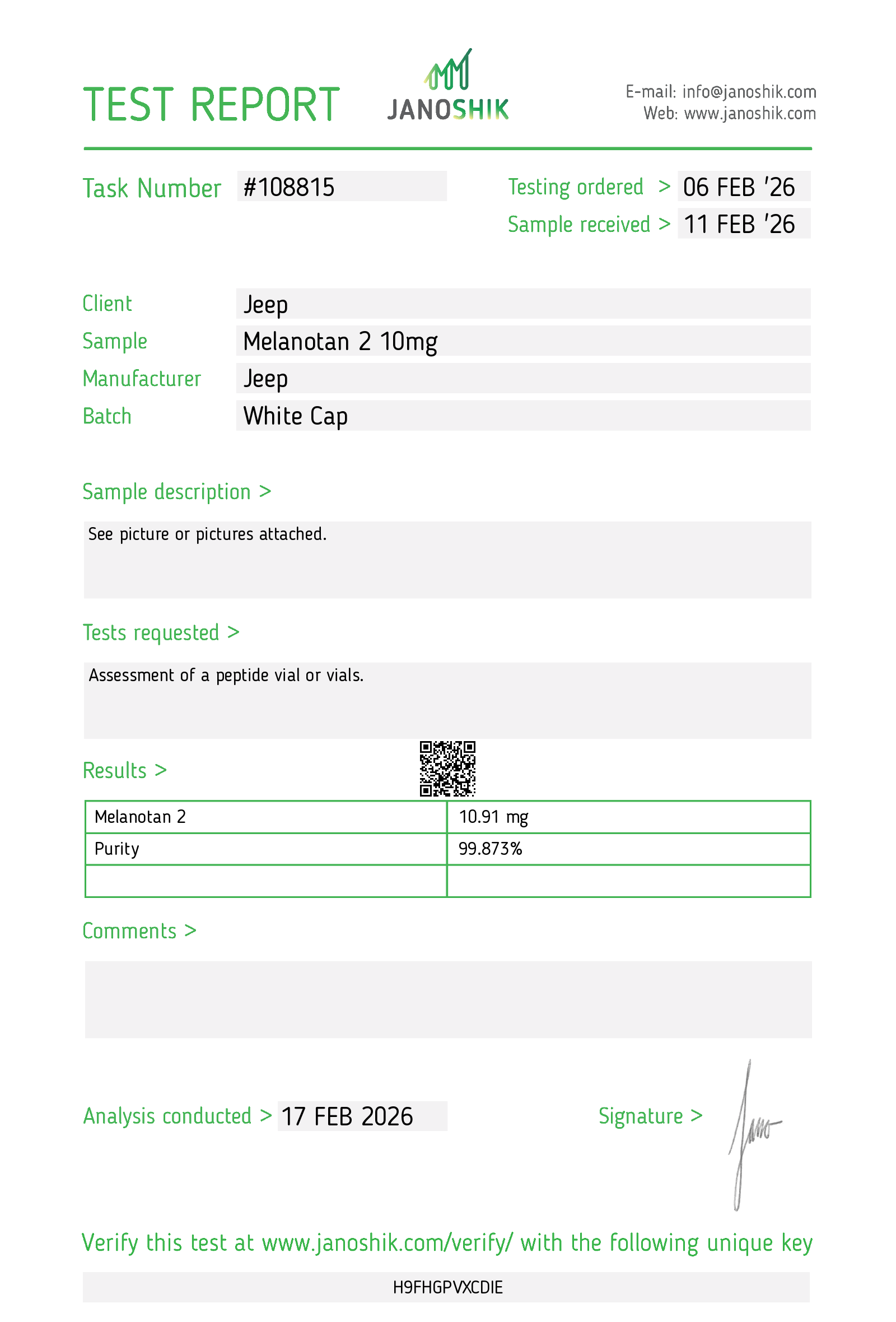
Task Number: #108612
Testing Ordered: 06 FEB ‘26
Sample Received: 11 FEB ‘26
| Field | Value |
|---|---|
| Client | Jeep |
| Sample | Bacteriostatic Water 3ml |
| Manufacturer | Jeep |
| Batch | White |
See picture or pictures attached.
Qualitative and quantitative analysis of non-AAS sample.
| Substance | Result |
|---|---|
| Benzyl Alcohol | 9.33 mg/ml |
(No comments provided)
16 FEB 2026
Signature: (Signed)
Verify this test at:
www.janoshik.com/verify/
Unique Key:
T4XMD1ZUBTYK
\u26a0\ufe0f This product is intended for in vitro research use only by qualified professionals. It is NOT approved for human or veterinary use. This document has not been evaluated by the FDA. SageBioLabs is not a compounding pharmacy. Purchaser assumes full responsibility for safe handling and legal compliance.
